Hint: Count the number of atoms of each element, and then multiply that number by the element's atomic weight. 
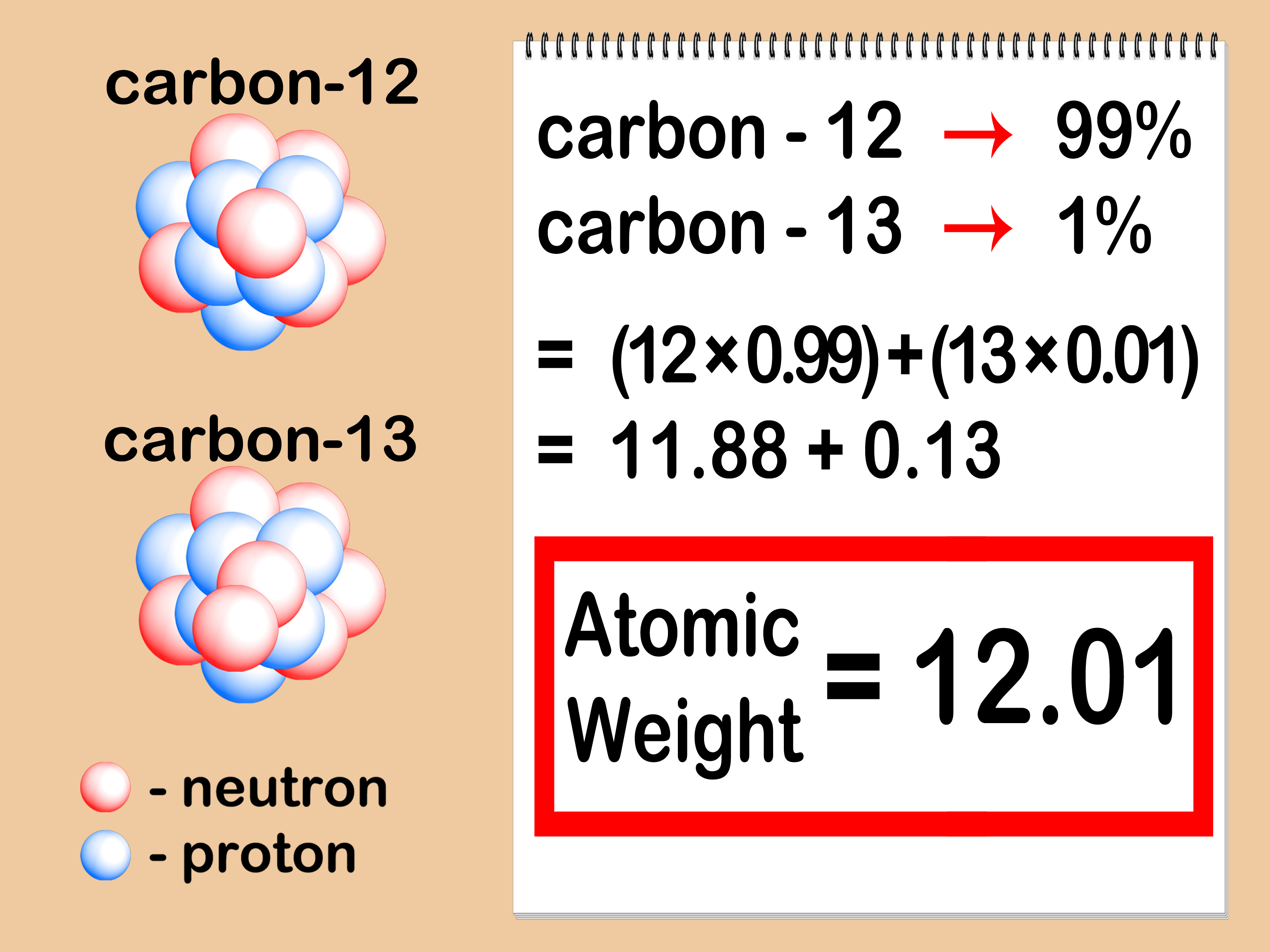
Use the table below to find the atomic weight of each atom (element), or refer to a Periodic Table of the Elements. Let us calculate the molecular weight of some common compounds. For example, in one mole of a chemical compound there are 6.022 x 1023 molecules. 2 Now, to calculate the molar mass of FeCl3, you just have to add the molar mass of all the individual atoms that are present in FeCl3. 1 The molar mass of Chlorine is 35.45 g/mol. You can see that The molar mass of Iron is 55.845 g/mol. Find out information on elements such as electronegativity, electron configuration, ionization energies Bond Type Predictor. So let’s look at the molar mass of Iron and Chlorine from the above periodic table. One mole of 'something' contains 6.022 x 1023 entities. Calculate the molar mass of any compound using a user-friendly and clickable periodic table Element Information. A mole is the unit that measures the amount of a substance. share my calculation Everyone who receives the link will be able to view this calculation. One thousand mers connected together would add up to a weight of 28,000 grams/mole and would have 6,000 atoms.Ī mole is the standard method in chemistry for communicating how much of a substance is present. Calculates the molar mass of the substance using atomic mass units from the periodic table All online. We combine (react) many mers of ethylene together to form a polyethylene chain. The atomic weight of carbon is 12 and that of hydrogen is 1, so one mer of ethylene has a weight of 2(12) + 4(1) = 28. It has a total of 6 atoms: 2 carbon (C) atoms and 4 hydrogen (H) atoms. The chemical formula for an ethylene monomer is -(CH2-CH2).The calculator takes the elemental composition of the compound and weighs the elements to get an empirical formula mass. In order to calculate the molecular weight of one water molecule, we add the contributions from each atom that is, 2(1) + 1(16) = 18 grams/mole. Our molar mass calculator uses the periodic table and the chemical formula to solve for the molar mass of a chemical compound based on the compounds empirical formula. Using the periodic table of the elements to find atomic weights, we find that hydrogen has an atomic weight of 1, and oxygen's is 16. The first step for calculating molar mass is to identify all the elements in a given molecule and write their atomic masses using the periodic table. The chemical formula for water is H2O, which means this molecule has 3 atoms: 2 of hydrogen (H) and 1 oxygen (O) atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
